18 March 2026
Reading time [minutes]: 22
Distributed Diagnostics
Decentralised diagnostics: operational and clinical efficiency
How point-of-care molecular diagnostics – thanks to room-temperature-stable multiplex PCR, portable devices such as bCUBE™ and cloud-based management via bAPP™ – tangibly improves clinical turnaround times and operational processes in healthcare.
Abstract
Purpose
To demonstrate, in measurable terms, how decentralised molecular diagnostics can improve clinical and operational efficiency within care pathways.[15]
Key technologies
Room-temperature-stabilised multiplex PCR kits.[1] Portable real-time PCR instruments and cloud-based software for managing results.[2]
Expected results
Diagnostic turnaround times have been reduced from hours or days to timeframes that allow for immediate clinical decision-making, thereby reducing the workload on the central laboratory and enabling more timely interventions.[4]
Impact
In hospital and community settings, the literature reports significant reductions in TAT.[5] Greater timeliness can have an impact on the appropriateness of treatment, isolation and the organisation of patient flows.[6]
- Snapshot
- Intro
- 1. Shorter waiting times in A&E, emergency departments and outpatient clinics
- 2. Less logistical and centralised overhead, greater operational continuity
- 3. Streamlined training, automated quality control and widespread deployment
- 4. Improved clinical outcomes thanks to timely diagnoses
- 5. KPIs and key indicators: TAT, cost per test, productivity per FTE
- 6. Regional and hybrid models: from hub-and-spoke to a widespread network
- 7. Conclusions
Snapshot
Ambient-stable multiplex PCR
PCR technologies capable of simultaneously detecting multiple pathogens in a single test, using reagents formulated to remain stable at room temperature. They eliminate the need for a cold chain, simplifying logistics and storage even outside the laboratory.[1]
bCUBE™
A miniaturised portable thermal cycler for real-time PCR developed by Helyx Industries S.p.A. – Hyris Division, part of the Hyris System™ platform.[2]
bAPP™
Cloud-based software platform for managing and analysing results from the Hyris System™.[2]
Turnaround Time (TAT)
This refers to the turnaround time of a diagnostic test, from the time the sample is taken until the results are available. A short turnaround time is crucial for timely clinical decisions, particularly in acute situations.[4]
Hub & Spoke
A ‘hub-and-spoke’ organisational model in which a central laboratory (hub) supports a network of peripheral testing sites (spokes). The most urgent tests can be carried out at the spokes, whilst the hub centralises specialist analyses, confirmatory tests or second-line processing, ensuring standardisation and quality.[12]
Intro
The ability to diagnose a condition rapidly is essential not only for treating patients promptly, but also for improving epidemiological surveillance and infection control.[3] During the COVID-19 pandemic, the limitations of a centralised approach became starkly apparent: relying exclusively on centralised laboratories led to delays in test results, with negative impacts on patient flow and the management of isolation.[5] Generally speaking, traditional core laboratories offer high throughput and standardisation, but suffer from their distance from the point of care: the time spent transporting samples and waiting for batches of analyses can erode the inherent speed advantage of techniques such as PCR.[4] This observation has driven the move towards decentralised diagnostics, the aim of which is to bring the test ‘to the patient’s bedside’ rather than the patient to the test. [4]
The molecular decentralisation solutions available today aim to combine the accuracy of laboratory diagnostics with the speed and accessibility of rapid tests. Within Helyx Industries S.p.A., the Hyris Division is developing a distributed qPCR ecosystem based on bCUBE™ and bAPP™. [2] In validated applications of the Hyris system published in the literature, results were obtained in under two hours without the need for a fully equipped laboratory.[13] In other words, molecular diagnostics can be made more distributed, connected and closer to the point of care, whilst maintaining a centralised approach to data and workflow management. [2]
In this article, we analyse how this decentralised approach tangibly improves clinical and operational efficiency. We will present use cases and key indicators (TAT, costs, productivity) by comparing the traditional model with the distributed one. In particular, we will examine: (1) the reduction in turnaround times in A&E, nursing homes and peripheral facilities; (2) the easing of the workload on centralised laboratories and sample logistics; (3) the impacts on staff training, quality control and operational continuity; (4) the clinical benefits in terms of outcomes thanks to more timely diagnoses; (5) some measurable KPIs; (6) hybrid regional organisational models, with a comparative table ‘traditional vs distributed’. The tone of voice remains predominantly technical (70%) to support laboratory and healthcare directors with data and literature, but with a strategic perspective (30%) useful for regional stakeholders, OEMs and investors.
Before going into detail, it is worth emphasising that decentralised diagnostics is no longer a futuristic concept. The 2022 ECDC mapping report documents the use of POCT devices for infectious diseases in various European countries and highlights their potential for surveillance, prevention and control.[3] In light of this, let us examine the operational and clinical benefits achieved by bringing the laboratory closer to the patient.
1. Shorter waiting times in A&E, emergency departments and outpatient clinics
Turnaround time (TAT) is perhaps the most important parameter for assessing the clinical efficiency of a diagnostic pathway. In traditional centralised workflows, samples taken in the A&E department or on the ward are sent to the laboratory, where they are often processed in batches, and the results are not available for many hours or even days. This delay can be critical in acute situations: patients with respiratory symptoms or fever must wait in isolation or be treated empirically, with risks of transmission and inappropriate use of antibiotics or antivirals.[6] Point-of-care molecular tests, by contrast, can drastically reduce TATs, bringing them in many cases down to around two hours and enabling a clinical decision to be made within the same care window.[4]
A landmark study in the field of COVID-19 (COV-19POC) has shown that the introduction of rapid point-of-care molecular tests in hospitals reduced the average turnaround time from 21.3 hours to 1.7 hours, a reduction of over 90%. [5] This translated into tangible benefits: fewer patients kept unnecessarily in isolation wards, faster allocation to the appropriate wards, and a reduction in length of stay for those testing negative. [5] Even outside a pandemic context, Drancourt et al. report that point-of-care laboratories can provide results within two hours, compared to one or two days for centralised laboratories, thereby improving clinical management and infection control.[4]
In the A&E department, a shorter turnaround time means being able to distinguish more quickly between bacterial and viral infections, identify specific pathogens, and initiate targeted treatments before the patient is admitted or discharged. The systematic review by Mojebi et al. (2024) highlighted that rapid molecular tests for respiratory viruses in A&E are associated with reduced antibiotic use, shorter isolation times and, in some studies, a reduction in hospital admissions.[6]
In care homes and long-term care facilities, rapid diagnosis is crucial for containing outbreaks. Residents are often frail, and a respiratory infection can spread rapidly, with significant clinical consequences. In a prospective study of residential settings, the implementation of point-of-care (POC) molecular testing for respiratory viruses enabled faster turnaround times and more timely outbreak declaration. [7] This enables immediate containment measures – isolation, contact tracing, and prophylaxis where indicated – thereby reducing the clinical and social impact.[7]
Even in local clinics and outlying healthcare centres, shorter response times improve the quality of service and patient management. A notable example comes from public programmes for the early diagnosis of HIV in infants in Africa: with point-of-care testing, same-day diagnosis and the faster initiation of antiretroviral therapy have become a tangible reality. [8] The principle is broader: when the result is available during the same consultation, the likelihood that the diagnosis will immediately lead to a useful clinical decision increases substantially.[15]
2. Less logistical and centralised overhead, greater operational continuity
In addition to turnaround times, decentralised diagnostics has a profound impact on the operational efficiency of healthcare systems. Centralised laboratories must manage high volumes, peaks in demand and the complex logistics of samples arriving from different locations. Each sample requires transport, tracking, registration and often a batch processing system to optimise the use of equipment. This creates logistical and administrative bottlenecks.[4]
Under a decentralised model, part of the workload is shifted directly to the point of care. Tests carried out in A&E or in local hospitals do not need to be transported, thereby reducing both time and logistics costs. Furthermore, the central laboratory can focus on highly complex analyses, confirmatory tests and specialist tests, thereby improving overall quality and productivity.[4]
Another key operational aspect is the robustness of the diagnostic workflow under less-than-ideal environmental conditions. In traditional scenarios, reagents and samples often need to be kept refrigerated during transport to prevent degradation. The stabilisation and lyophilisation of PCR reagents, however, reduces reliance on the cold chain and simplifies transport, storage and availability of kits outside the centralised laboratory. [1] In a study of a ready-to-use freeze-dried multiplex master mix, the formulation proved stable with no loss of analytical performance even after exposure to temperature fluctuations during transport.[1] For a distributed platform, this means reduced logistical vulnerability and greater operational continuity at peripheral sites.[1]
Round-the-clock operational continuity is a further benefit. Centralised laboratories often have limited opening hours or require costly night shifts to ensure continuous service. POC devices, on the other hand, can be integrated into extended operating models, enabling faster diagnoses even at night or at weekends.[4]
3. Streamlined training, automated quality control and widespread deployment
A common objection to decentralised diagnostics is the need for specialist staff. Traditionally, PCR requires technical expertise, sample preparation and the interpretation of results. However, technological advances have reduced these requirements thanks to portable systems and integrated workflows, designed to combine speed, simplicity and operational robustness. [9] This allows operations to be extended to non-specialist staff, provided they are adequately trained and operate within robust quality procedures.[10]
Helyx Industries has implemented this approach in an integrated platform based on bCUBE™ and bAPP™.[2] In the validation studies published on the Hyris system for SARS-CoV-2, results were reported more quickly than with reference methods and the system proved suitable for use outside a fully equipped laboratory.[13]
In terms of quality control, modern molecular decentralisation relies on standardised protocols, internal checks and connectivity with the central laboratory.[4] In decentralised models involving task shifting, test quality can remain comparable even when part of the work is carried out by non-laboratory staff, provided the system is supported by adequate training and governance.[10]
In validated systems, the analytical performance of POC molecular tests can be comparable to that of reference methods, with the advantage of a significantly shorter turnaround time.[13] The result is greater operational scope: more sites and more staff involved, but all operating under a single, centrally coordinated quality framework.
4. Improved clinical outcomes thanks to timely diagnoses
Clinical efficiency is not measured solely in terms of minutes saved, but in terms of improved outcomes. In clinical microbiology, access to a rapid and actionable diagnosis can influence treatment decisions to a far greater extent than the cost of the test itself. [15] In the case of patients with respiratory symptoms, the review by Mojebi et al. (2024) highlighted that rapid molecular tests can reduce the inappropriate use of antibiotics and improve clinical management, particularly when integrated into stewardship programmes.[6]
In the case of serious infections such as sepsis or meningitis, every hour counts: delays in diagnosis can lead to suboptimal empirical treatment or delays in starting targeted therapy, potentially worsening outcomes.[15] Although this article uses respiratory infections as a case in point, the logic applies across the board: reducing TAT means bringing forward critical clinical decisions.[15]
In the context of care homes and long-term care, rapid diagnosis enables the prompt isolation of those who test positive and helps prevent outbreaks. This reduces not only potential mortality but also the social and organisational impact associated with managing outbreaks in vulnerable populations.[7]
5. KPIs and key indicators: TAT, cost per test, productivity per FTE
To assess the effectiveness of decentralised diagnostics in practical terms, it is useful to consider a number of measurable KPIs.
Average turnaround time (TAT): as previously noted, molecular point-of-care (POC) tests can reduce TAT from over 20 hours to less than 2 hours in hospital settings.[5] In many cases, the reduction can exceed 90%, transforming clinical decisions that previously took a day into decisions made during the same consultation or hospital visit.[5]
Cost per test: assessing the ‘full’ cost of a diagnostic test requires taking into account not only reagents and staff, but also logistics, length of hospital stay, isolation, clinical appropriateness and turnaround time. From this perspective, modelling studies on certain molecular applications show that POC testing can be cost-effective and, in specific scenarios, even cost-saving compared to a centralised model. [11] The key point is that the unit cost of the reagent does not account for the full cost of the diagnostic pathway.[15]
Productivity per FTE (Full-Time Equivalent): decentralisation can increase the system’s overall capacity to deliver tests by distributing the workload between peripheral sites and the central laboratory. Automation and standardisation of workflows reduce manual steps and allow part of the activity to be shifted to peripheral sites, leaving the central laboratory to handle more complex analyses. [4]
From an organisational perspective, decentralisation also makes it easier to address the chronic shortage of laboratory staff. Instead of concentrating all demand in a single hub, the system can distribute part of the workload across departments and regions, whilst keeping governance, quality and specialist validation at the centre. This does not replace the central laboratory, but utilises it more efficiently.
6. Regional and hybrid models: from hub-and-spoke to a widespread network
Decentralised diagnostics can be implemented using a variety of organisational models. There is no single one-size-fits-all approach; often, the best solution is a hybrid model, which combines point-of-care testing with a central reference laboratory.
A common model is the hub-and-spoke system: a central laboratory (the hub) oversees and supports peripheral sites (the spokes) that carry out the most urgent tests. Within a national or regional diagnostic network, this model can extend geographical coverage whilst reducing infrastructure investment and the fragmentation of expertise.[12]
Another model is total distribution, in which every healthcare facility has molecular diagnostic capabilities. This approach is useful in highly decentralised settings or in emergency situations, but requires greater coordination, data governance and standardisation of processes.[12]
Comparison of the traditional diagnostic model versus the distributed model
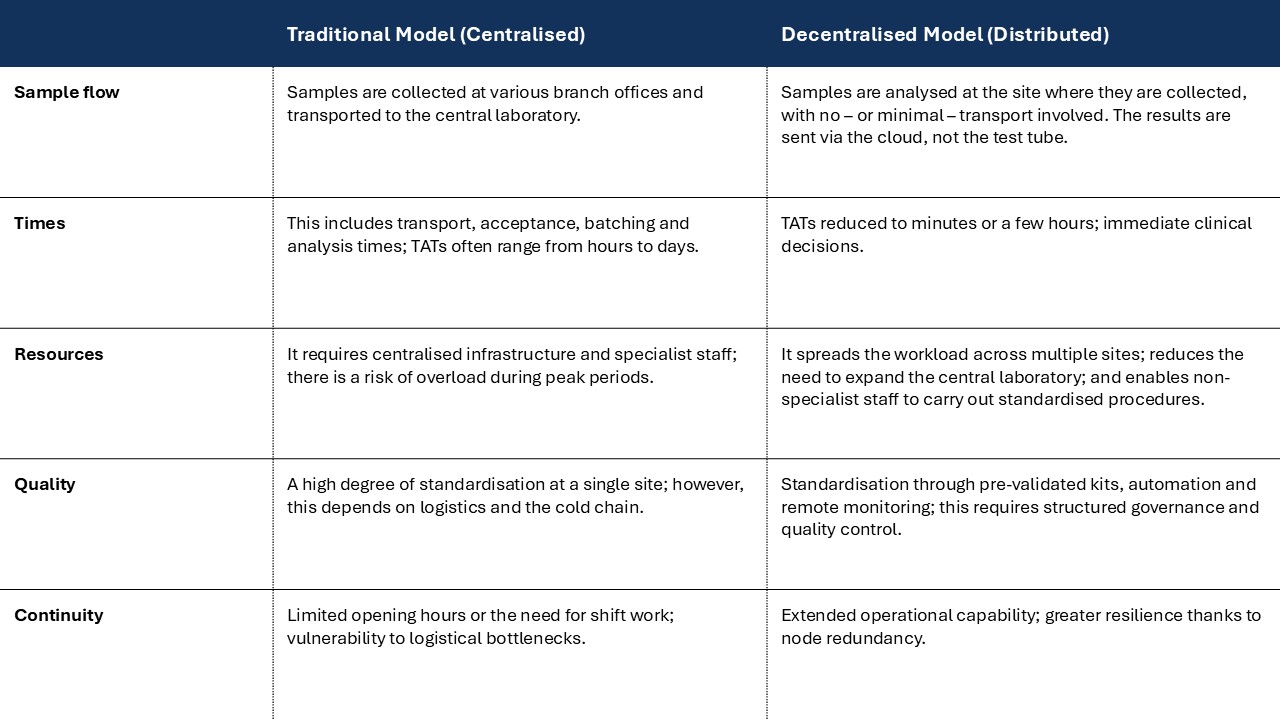
(The hybrid hub-and-spoke model combines elements of both: transport is reduced but not eliminated; point-of-care testing is carried out for urgent cases, whilst hubs handle confirmatory testing or high-volume testing; local staff carry out initial screening, and a central hub maintains standards and manages complex cases.)
7. Conclusions
Decentralised molecular diagnostics is now one of the most promising innovations for combining clinical effectiveness with operational efficiency. Through stabilised reagents, portable PCR devices and management software, advanced molecular diagnostics can be brought closer to the point of care.[1] In validated systems, this approach has been associated with faster turnaround times and better alignment between diagnosis and clinical decision-making. [13]
The data show that it is not simply a matter of speeding up testing, but of translating speed into better outcomes: less unnecessary waiting, more appropriate treatment and more timely interventions. In the COV-19POC study, for example, the reduction in the average turnaround time from 21.3 hours to 1.7 hours had a direct impact on patient allocation and the management of isolation.[5]
From an operational and managerial perspective, decentralisation reduces the burden on large laboratories and makes peripheral facilities more responsive. This distribution of diagnostic capacity increases the system’s resilience, particularly when demand rises rapidly or when logistical constraints make it difficult to rely solely on the central laboratory.[3]
Of course, every innovation brings its own challenges: implementing decentralised diagnostics requires initial investment, widespread training, IT integration and a shift in mindset. Clear governance must be established, quality controls must be guaranteed, and it must be ensured that the data produced is reliable and integrated into information systems. However, the available literature shows that organisational barriers can be overcome through appropriate models of standardisation, supervision and networking.[4]
From a strategic perspective, adopting decentralised diagnostic solutions has become a key factor in competitiveness and sustainability for many healthcare organisations. A hospital that reduces diagnosis times and optimises the clinical pathway improves the quality of care; a regional healthcare system that distributes diagnostic capacity becomes more resilient and ensures fairer access to care.
With “Decentralised Diagnostics: Operational and Clinical Efficiency”, we aimed to provide a comprehensive overview – including data, examples and organisational models – of how this development is having a positive impact on both healthcare management and patients’ lives. Molecular decentralisation is not merely a technology: it is a paradigm shift that is accelerating medical progress and reshaping the organisation of diagnostic services.
In the future, we can expect further advances: the integration of more sophisticated artificial intelligence to predict outbreaks or assist in interpreting results, the expansion of multiplex testing menus, and the roll-out of increasingly widespread diagnostic network models. In conclusion, decentralising diagnostics means accelerating medicine. Operational and clinical efficiency can finally go hand in hand.
Sources and Bibliography
[1] Nagaraj S, Ramlal S, Kingston JJ, et al. Thermostabilization of indigenous multiplex polymerase chain reaction reagents for detection of enterotoxigenic Staphylococcus aureus. J Microbiol Immunol Infect. 2018;51(2):191-198. DOI: 10.1016/j.jmii.2016.04.004. PubMed: https://pubmed.ncbi.nlm.nih.gov/27260780/
[2] Martinelli F, Ciccozzi M, Al Moghazi S, et al. Application of a portable instrument for rapid and reliable detection of SARS-CoV-2 infection in any environment. Immunol Rev. 2020;295 Suppl 1:4-10. DOI: 10.1111/imr.12857. PubMed: https://pubmed.ncbi.nlm.nih.gov/32329102/
[3] ECDC. Assessment of point-of-care testing devices for infectious disease surveillance, prevention and control – a mapping exercise. 2022. https://www.ecdc.europa.eu/en/publications-data/assessment-point-care-testing-devices-infectious-disease-surveillance-prevention
[4] Drancourt M, Michel-Lepage A, Boyer S, Raoult D. The point-of-care laboratory in clinical microbiology. Clin Microbiol Rev. 2016;29(3):429-447. DOI: 10.1128/CMR.00090-15. PubMed: https://pubmed.ncbi.nlm.nih.gov/27029593/
[5] Brendish NJ, Poole S, Naidu VV, et al. Clinical impact of molecular point-of-care testing for suspected COVID-19 in hospital (COV-19POC): a prospective, interventional, non-randomised, controlled study. Lancet Respir Med. 2020;8(12):1192-1200. DOI: 10.1016/S2213-2600(20)30454-9. PubMed: https://pubmed.ncbi.nlm.nih.gov/33038974/
[6] Mojebi A, Wu P, Keeping S, et al. Clinical impact of rapid molecular diagnostic tests in patients presenting with viral respiratory symptoms: a systematic literature review. PLoS One. 2024;19(6):e0303560. DOI: 10.1371/journal.pone.0303560. PubMed: https://pubmed.ncbi.nlm.nih.gov/38870136/
[7] Tan C, Chan CK, Ofner M, et al. Implementation of point-of-care molecular testing for respiratory viruses in congregate living settings. Infect Control Hosp Epidemiol. 2024;45(9):1085-1089. DOI: 10.1017/ice.2024.72. PubMed: https://pubmed.ncbi.nlm.nih.gov/38659123/
[8] Boeke CE, Joseph J, Wang M, et al. Point-of-care testing can achieve same-day diagnosis for infants and rapid ART initiation: results from government programmes across six African countries. J Int AIDS Soc. 2021;24(3):e25677. DOI: 10.1002/jia2.25677. PubMed: https://pubmed.ncbi.nlm.nih.gov/33745234/
[9] Bissonnette L, Bergeron MG. Portable devices and mobile instruments for infectious diseases point-of-care testing. Expert Rev Mol Diagn. 2017;17(5):471-494. DOI: 10.1080/14737159.2017.1310619. PubMed: https://pubmed.ncbi.nlm.nih.gov/28343420/
[10] Simmonds FM, Cohn J, Moyo C, et al. Task shifting for point-of-care early infant diagnosis: a comparison of the quality of testing between nurses and laboratory personnel in Zimbabwe. Hum Resour Health. 2020;18(1):4. DOI: 10.1186/s12960-020-0449-2. PubMed: https://pubmed.ncbi.nlm.nih.gov/31992332/
[11] le Roux SM, Dramowski A, Finlayson H, et al. Cost-effectiveness of point-of-care versus centralised, laboratory-based nucleic acid testing for diagnosis of HIV in infants: a systematic review of modelling studies. Lancet HIV. 2023;10(5):e320-e331. DOI: 10.1016/S2352-3018(23)00029-2. PubMed: https://pubmed.ncbi.nlm.nih.gov/37149292/
[12] Mataka A, Tumbare EAJ, Holtzman D, et al. Strategic site selection for placement of HIV early infant diagnosis point-of-care technology within a national diagnostic network in Lesotho. Afr J Lab Med. 2021;10(1):1156. DOI: 10.4102/ajlm.v10i1.1156. PubMed: https://pubmed.ncbi.nlm.nih.gov/34522629/
[13] Miscio L, Olivieri A, Labonia F, et al. Evaluation of the diagnostic accuracy of a new point-of-care rapid test for SARS-CoV-2 virus detection. J Transl Med. 2020;18(1):488. DOI: 10.1186/s12967-020-02651-y. PubMed: https://pubmed.ncbi.nlm.nih.gov/33349261/
[14] Padoan A, Cosma C, Aita A, et al. Hyris bCUBE SARS-CoV-2 rapid molecular saliva testing: a POCT innovation on its way. Clin Chem Lab Med. 2022;60(5):766-770. DOI: 10.1515/cclm-2022-0008. PubMed: https://pubmed.ncbi.nlm.nih.gov/35041302/
[15] Hansen GT. Point-of-Care Testing in Microbiology: A Mechanism for Improving Patient Outcomes. Clin Chem. 2020;66(1):124-137. DOI: 10.1373/clinchem.2019.304782. PubMed: https://pubmed.ncbi.nlm.nih.gov/31811002/